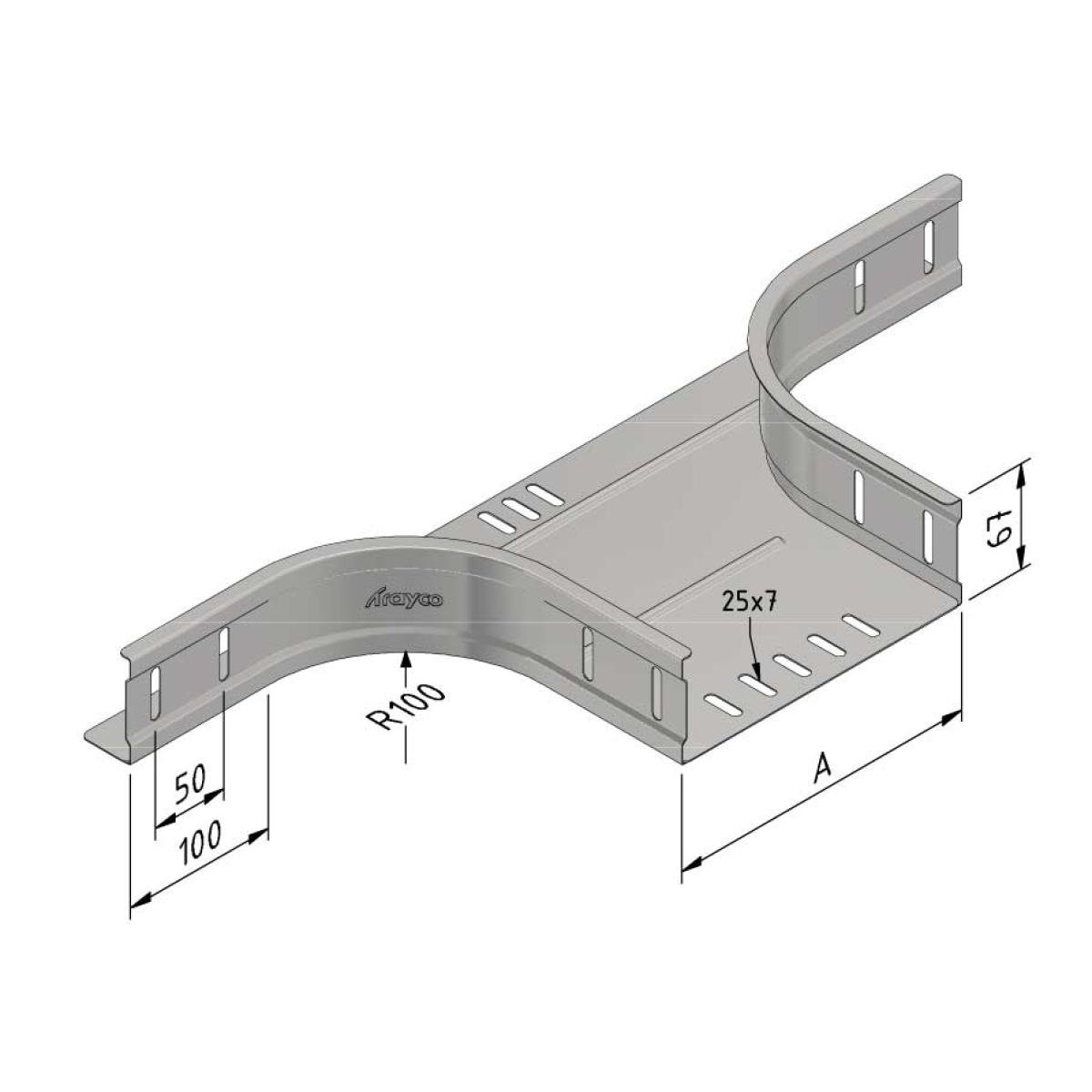
Cable Tray Branch
SS-CT-BR_
Cable Tray Branch
SS-CT-BR_
| SKU | Article code | Finishing | Dimension A | Packaging | |||
|---|---|---|---|---|---|---|---|
|
|
13387 |
CT60-BR-100-SS316 |
SS316
|
100
|
1
|
Default
|
|
|
|
13389 |
CT60-BR-200-SS316 |
SS316
|
200
|
1
|
Default
|
|
|
|
13390 |
CT60-BR-300-SS316 |
SS316
|
300
|
1
|
Default
|
|